Credibility Assessment of Models
How to build a model we can trust? Established regulatory-aligned verification and validation (V&V) frameworks (as per ASME V&V 40) with a focus on Identifiability, Uncertainity Quanification and Sensitivity Analysis.
This project is part of my post-doctoral research activities with the Division of Biomechanics at Norwegian University of Science and Technology (NTNU), Trondheim, Norway. I am planning to write long format essay or white paper on “How to build models that we can trust?”
The following is the summary for a quick reference. I am also drafting a manuscript, whcih will as act as guideline and an example on how to use the [ASME V&V 40-2018](https://www.asme.org/codes-standards/find-codes-standards/assessing-credibility-of-computational-modeling-through-verification-and-validation-application-to-medical-devices - Assessing Credibility of Computational Modeling through Verification and Validation: Application to Medical Devices for the compuational model that you are developing.
More details to be updated soon.
Introduction
This page documents a complete guide on how to build a computational model we can trust, using an evidence-based, regulatory-aligned approach grounded in ASME V&V 40 guidelines. The goal is to establish a structured 18-step workflow that ensures credibility across the model lifecycle, from initial context definition to final verification and validation.
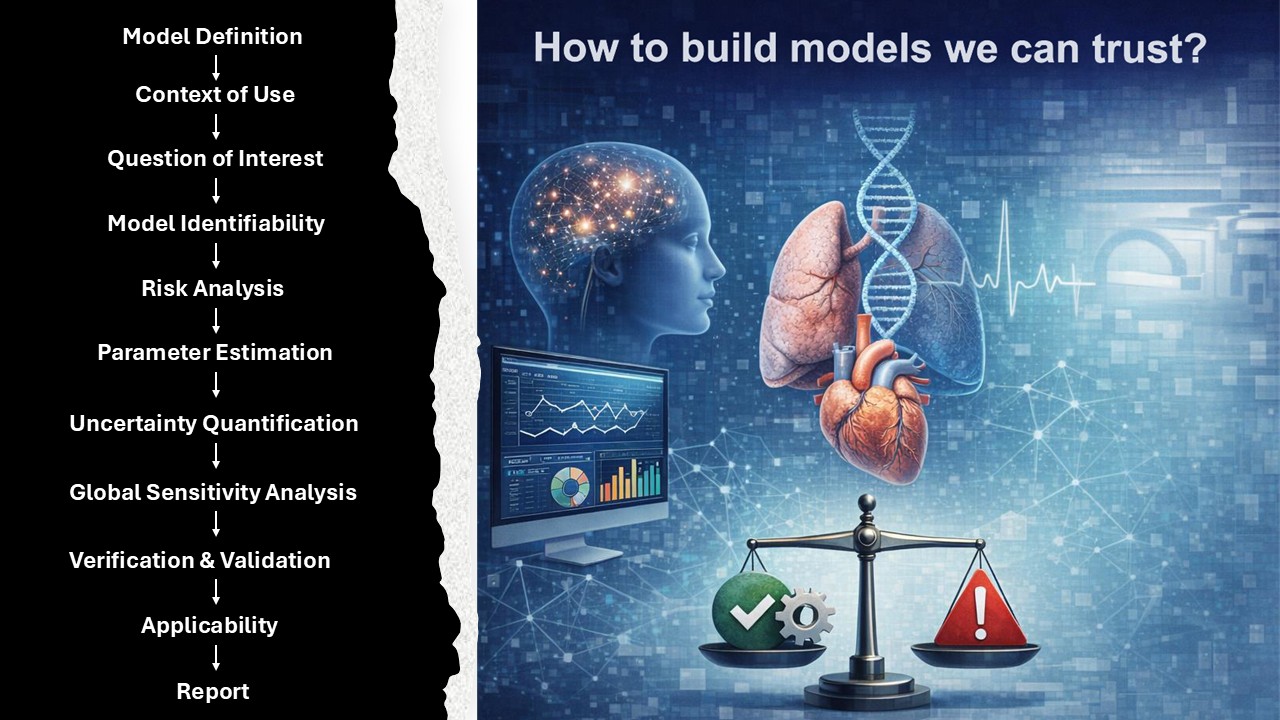
Workflow Overview
Step 1: Question of Interest (QoI)
What do we want to know?
- Define the primary scientific or clinical question.
- Ensure clarity in the scope and measurable objectives.
Step 2: Model Definition
- Mathematical formulation:
- Equations
- States
- Parameters
- Explicitly state assumptions supporting model structure.
Step 3: Structural Identifiability
- Can model parameters be uniquely estimated assuming ideal, noise-free data?
Step 4: Observability
- Are model outputs inferable from measurable signals/outputs?
Step 5: Controllability
- Can system outputs be influenced by altering inputs (e.g., preload, afterload)?
- Critical for drug/therapy response simulation and feedback control models.
Step 6: Screening Sensitivity Analysis
- Initial sensitivity check to identify which parameters matter.
Step 7: Practical Identifiability
- Perform profile likelihood analysis incorporating real experimental/clinical data and noise constraints.
Step 8: Context of Use (CoU)
- Define intended application and decision supported by the model.
Step 9: Risk Analysis
- Risk = model influence × consequence of wrong decision
- Apply ASME V&V 40 risk-based rigor for downstream steps.
Step 10: Parameter Estimation
- Employ robust estimation techniques validated for the given data and noise level.
Step 11: Hyperparameter Tuning
- Optimize tuning parameters for model selection or machine learning-based sub-models.
Step 12: Uncertainty Quantification (UQ)
- Quantify aleatory & epistemic uncertainties:
- Monte Carlo
- Polynomial Chaos
- Bayesian Inference
Step 13: Global Sensitivity Analysis
- Identify global influencers using:
- Sobol indices
- Morris method
- Variance decomposition
Step 14: Verification
- Ensure:
- Correctness of code
- Numerical accuracy
- Unit & integration tests
Step 15: Validation
- Compare model predictions to independent validation data:
- Use quantitative metrics & visual analysis
- Assess prediction intervals
Step 16: Applicability
- Check:
- Validation-Context overlap
- Relevance of predictions to original QoI
Step 17: Explainability
- Ensure transparency of:
- Parameter influence
- Model decision pathways
- Use interpretable representations for regulatory and scientific acceptance.
Step 18: Reporting
- Provide:
- Complete traceability of assumptions
- Verification, Validation, UQ, SA evidence
- A Credibility Matrix summarizing strength-of-evidence vs risk
Best Practices
- Validate QoI alignment before investing in complexity.
- Use identifiability checks before parameter estimation.
- Always report Credibility Matrix with context-specific rigor.
- Align every step with regulatory science recommendations.